Copper sulfide minerals play a crucial role in the global mining industry due to the extensive use of copper in various applications ranging from electrical wiring to construction materials. Understanding the floatability of these minerals is essential for efficient extraction processes. This article delves into the main copper sulfide minerals, their floatability characteristics, and the copper flotation process.
Copper Sulfide Minerals Floatability
Main Copper Sulfide Minerals and Their Floatability
Chalcopyrite
Chalcopyrite (CuFeS2) contains Cu34.57% and is the main copper mineral. Chalcopyrite can maintain its natural floatability for a long time in neutral and weakly alkaline media. Still, in strongly alkaline (pH>10) media, an iron hydroxide film is formed due to the surface structure being corroded by OH-; its natural floatability decreases. The chalcopyrite on the deposit’s surface has been oxidized for a long time, its hardness has become smaller, and it is easy to be crushed, so its floatability has deteriorated.
The most commonly used collectors for flotation chalcopyrite are xanthate and black powder. In recent years, it has also used sulfur nitrogen and thiamine esters. Some use thiourea salt, butyl acrylate, etc., to replace xanthate flotation chalcopyrite in foreign countries.
Chalcopyrite is easily inhibited by cyanide and oxidants in alkaline media. For example, in the separation of copper and lead, cyanide is commonly used to inhibit chalcopyrite; in the separation of copper and molybdenum, oxidants have been widely used to inhibit chalcopyrite. Copper salts (such as copper sulfate) sometimes activate suppressed chalcopyrite.
Chalcocite
Chalcocite (Cu2S) contains 79.8% Cu, the most common secondary copper sulfide mineral. It is brittle and easily sludged by over-crushing.
The copper mineral in many large porphyry copper mines abroad is chalcocite. The collector of chalcocite is mainly xanthate. It has good floatability in both acidic and alkaline media. Because the lattice energy of copper-sulfur crystals in chalcocite is smaller, the radius of copper ions is small, and the radius of sulfur ions is large, it is easy to be exposed to oxidation, so chalcocite is easier to oxidize than chalcopyrite. After oxidation, more copper ions enter the pulp. The presence of these copper ions will activate other minerals or consume chemicals, making sorting difficult.
The inhibitors of chalcocite are Na2S03, Na2S2O3, K3Fe (CN) 6, and K4Fe (CN) 6. A large amount of Na2S also has an inhibitory effect on chalcocite. The inhibitory effect of cyanide on chalcocite is weak because copper ions on the surface of chalcocite continue to dissolve and interact with cyanide, rendering it ineffective. Only by continuously adding cyanide can the suppression purpose be achieved.
Bornite
The chemical composition of bornite (Cu5FeS4) is not fixed. According to the molecular formula, it contains Cu63.3%. There are two types: primary and secondary. The surface properties and floatability of bornite are between chalcocite and chalcopyrite. When xanthate is used as a collector, it can float in acidic and weakly alkaline media. When the pH is >10, its floatability decreases. In strongly acidic media, its floatability also deteriorates significantly and is efficiently inhibited by cyanide.
Other copper sulfide minerals
Other copper sulfide minerals, such as copper blue (CuS), have similar floatability to chalcocite. Arsenite 3Cu2S·As2S3 is a primary copper ore. It is an equiaxed crystal system and does not dissociate. It has many isomers. It has low hardness and high brittleness and can easily become muddy due to over-grinding. When flotation of arsenic ore with xanthate is used, the most suitable pH is 11 to 12. Using sodium carbonate rather than lime as a medium adjuster is better because it inhibits arsenic tetrahedrite when free CaO is higher than 400g/m3. When the sodium sulfide dosage is low (30 mg/L), the flotability can be improved due to the sulfidation of the oxidized surface. However, increasing the dosage can completely inhibit the flotation of arsenite.
Floatability rules of copper sulfide minerals
Regarding the floatability of copper sulfide minerals, the following rules can be summarized:
- All minerals that do not contain iron, such as chalcocite and covellite, have similar floatability, and the inhibitory effect of cyanide and lime on them is weak.
- All iron-containing copper minerals, such as chalcopyrite, bornite, etc., are efficiently inhibited by cyanide and lime in alkaline media.
- The xanthate collector mainly chemically adsorbs the cationic Cu2+, so minerals containing more Cu2+ on the surface strongly affect the xanthate. The order of the strength of the effect is chalcocite > copper blue > bornite > chalcopyrite.
- The floatability of copper sulfide minerals is also affected by crystal particle size, embedded particle size, and primary and secondary factors. If the crystals and embedded fabrics are too fine, it will be more difficult to float. Secondary copper sulfide ores are easily oxidized and more challenging to float than primary copper ores.
Copper Sulfide Beneficiation
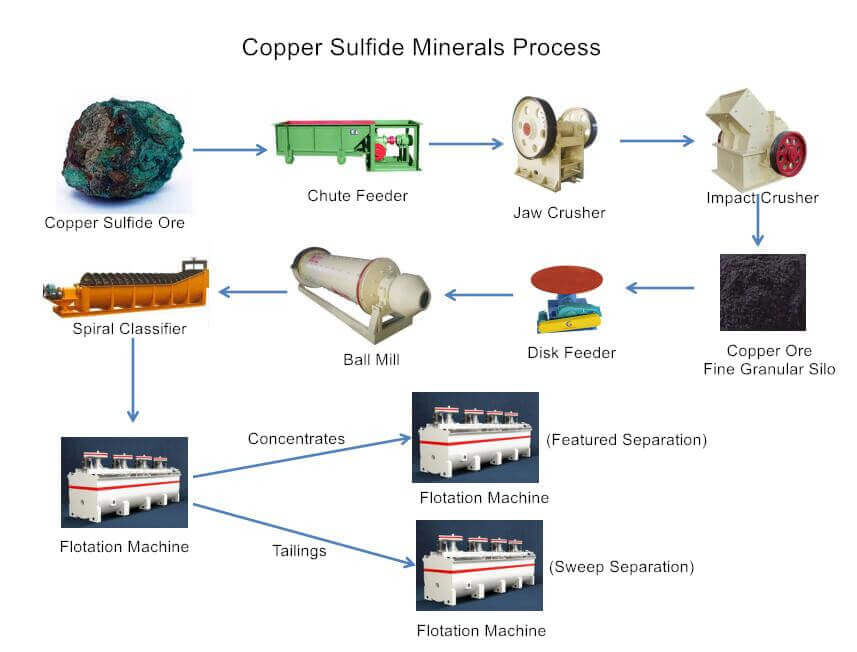
The main beneficiation process of copper sulfide ore is flotation. According to the embedded characteristics of different copper sulfide ores, it can be divided into a one-stage grinding + flotation process, one-stage grinding + flotation + coarse concentrate regrinding process, and two-stage grinding + two-stage (one-stage) flotation process.
1. One-stage grinding + flotation process
This process is suitable for copper sulfide ores where the copper and gangue minerals are loosely associated, and the embedded particle size is coarse and uniform.
Copper sulfide ore can achieve better monomer dissociation effects after one grinding process and can directly enter the flotation process for separation. Generally, it can be achieved through one rough separation, one to three featured separations, and one sweep, which can get a better flotation indicator.
The one-stage grinding + flotation process of copper sulfide is simple and easy to operate, and the mineral processing cost is low.
2. One-stage grinding + flotation + coarse concentrate regrinding process
This process suits single sulfide or copper-molybdenum ore from the Banyan copper mine.
After primary grinding, the copper sulfide ore enters the flotation process for roughing and sweeping. This process discards a large amount of tailings. The obtained coarse concentrate contains a large amount of concatenation of copper and associated minerals such as quartz, resulting in a poor concentrate grade. It is not high, so it needs secondary grinding for monomer dissociation and then two to three times of beneficiation to finally obtain copper concentrate.
The concentrate quality of the copper sulfide one-stage grinding + flotation + coarse concentrate regrinding process is higher, and the mineral processing index is also better.
3. Two-stage grinding + two-stage (one-stage) flotation process
This process is suitable for copper sulfide ores with uneven particle size.
After the copper sulfide ore is coarsely ground for the first time, it can be flotated to select some coarse-grained copper minerals. After a second grinding to enhance the dissociation of the mineral monomers, fine-grained copper can be flotated.
The two-stage copper sulfide grinding + two-stage (one-stage) flotation process can recover useful minerals to the greatest extent.
The copper sulfide flotation process mainly consists of the above three methods, which are more suitable for processing copper sulfide ores with a single mineral composition. Through mineral processing experiments, we can determine the specific mineral processing technology and process for more complex copper sulfide ores.
As advancements in mining technology continue to evolve, a deeper understanding of the floatability of copper sulfide minerals will undoubtedly contribute to optimizing flotation processes and the sustainability of the copper mining industry. These minerals can be selectively separated from gangue materials through flotation, producing high-grade copper concentrates.
LATEST PRODUCTS
-
Tubular Screw Conveyor
【Capacity】6-50 m3/h 【Procesible Material】 …
-
Heavy Plate Feeder
Capacity: 100-240 m3/h Power: 15-45 kW Speed: 0…
-
Plate Magnetic Separator
【Capacity】8-35 t/h 【Power】1.5-3 kW 【Applic…