The various agents used in the flotation process are collectively referred to as flotation agents. In the flotation agents, except for collectors and foaming agents, they are all called regulators. The role of the regulator is to adjust the role of the collector and the mineral, promote or inhibit the float-ability of the mineral; adjust the pH of the pulp and the composition of ions. According to the effect of regulators, it can be roughly divided into pH regulators, activators, inhibitors, dispersants, flocculants, etc. In the last article we introduce pH regulators, activators, dispersants, and flocculants, today we will introduce the inhibitor to you.
Inhibitor
Commonly used inhibitors are sodium sulfide, zinc sulfate, sodium cyanide, potassium dichromate, water glass, lime, yellow blood salt, tannin, starch (dextrin), carboxymethyl cellulose, etc.
1. Sodium Sulfide
Sodium sulfide is the activator of non-ferrous metal oxide ore, and when the addition amount is large enough, it is also an inhibitor of sulfide ore.
The preparation of sodium sulfide is to reduce sodium sulfate (Na2SO4) by burning coal, charcoal, etc. as a reducing gas.
The reaction formula is: Na2SO4+2C=Na2S+2CO2↑
Sodium sulfide is used as an inhibitor of sulfide ore in flotation operations. In the production practice of molybdenum separation, sodium sulfide is used to inhibit pyrite, and kerosene is used as a collector to flotation molybdenite. The buoyancy is good and is not inhibited by sodium sulfide. Sodium sulfide inhibits pyrite. After several selections, a qualified molybdenum concentrate is obtained.
When sodium sulfide is added to the pulp, the pulp is alkaline, which makes the surface of the sulfide mineral form a layer of hydrophilic hydroxide compound and becomes hydrophilic, so that the sulfide mineral is inhibited.
2. Zinc Sulfate
Zinc sulfate is prepared by the action of zinc scraps from metal processing plants and dilute sulfuric acid. Zinc sulfate is an inhibitor of zinc blende, and its effect is not very obvious when used alone. It has a strong inhibitory effect when used with alkali, sodium cyanide, sodium sulfite, etc. The higher the pH value of the slurry, the better the inhibitory effect.
3. Sodium Cyanide (Potassium)
When the priority flotation process is adopted for polymetallic deposits, sodium cyanide is used to inhibit sulfide minerals such as pyrite, sphalerite, chalcopyrite, etc. The mixed-use of sodium cyanide and zinc sulfate has a very good inhibitory effect on sphalerite. When the amount of sodium cyanide is small, pyrite can be suppressed, and when the amount is larger, sphalerite can be suppressed, and when the amount is increased, various copper sulfide minerals can be suppressed.
In production practice, due to the toxicity of sodium cyanide, sulfur dioxide or sodium sulfite is often used instead. Sulfur dioxide and sodium sulfite have weaker inhibitory effects than sodium cyanide, but due to their low toxicity and easy oxidation by air, wastewater is easy to treat and is often used. Another advantage is that minerals inhibited by sulfur dioxide and sodium sulfite are more easily activated by copper sulfate, while minerals inhibited by sodium cyanide are more difficult to activate.
4. Lime
Lime’s inhibition of pyrite: Lime’s inhibition of pyrite is due to the formation of a hydrating film of calcium sulfate, calcium carbonate, and calcium oxide on its surface.
To activate the pyrite inhibited by lime, sodium carbonate and copper sulfate can be used, or sulfuric acid can be added to reduce the pH value of the pulp to 6-7, and butyl xanthate can be added to float the pyrite.
5. Phosphorus
Phosphorus Knox is prepared from phosphorus pentasulfide and sodium hydroxide as raw materials. The sodium hydroxide is formulated into a 10% aqueous solution, and then phosphorus pentasulfide is added. After stirring for 20 minutes, dilute the prepared solution to 0.5% to 1%, and then put it into use. The ratio of sodium hydroxide to phosphorus pentasulfide is 1:1.
6. Water Glass
Water glass is an inorganic colloid and is the most commonly used inhibitor in flotation operations. Water glass has a good inhibitory effect on quartz, silicate minerals, and aluminosilicate minerals (such as mica, feldspar, garnet, etc.), and is widely used as an inhibitor of gangue.
Water glass is a water glass sintered block formed by heating and melting quartz sand and sodium carbonate. The sintered block is dissolved in water to form a paste-like colloid. Its composition is complex, containing sodium metasilicate Na2SiO3, sodium orthosilicate Na2SiO4, sodium metasilicate Na2SiO5, and SiO2 colloidal particles. Commonly used Na2SiO3 said.
Quartz and sodium carbonate are used to make water glass. The properties of water glass are different due to the different preparation ratios of the application materials. The ratio of Na2O and SiO2 is generally used to express the composition of water glass. The ratio of mNa2O·nSiO2 n/m is called water glass. The modulus of the water glass used for floating, the modulus n/m=2.0~3.0, the modulus of the commonly used water glass quality standard is 2.2. The water glass with a small modulus has strong alkalinity, and the water with a large modulus is difficult to dissolve and has a strong inhibitory effect.
The inhibitory effect of water glass is mainly HSiO3- and H2SiO3, silicic acid molecule H2SiO3, and silicate ion HSiO3-has strong hydration, and it is a kind of colloidal particles and ions with strong hydrophilicity. HSiO3- and H2SiO3 are combined with Silicate minerals that have the same acid radicals and are easy to adsorb on the surface of quartz and silicate minerals to form a hydrophilic film, which increases the hydrophilicity of the mineral surface and suppresses it.
7. Sodium Thioglycolate
Sodium thioglycolate is used for molybdenum beneficiation operations. When the copper content of molybdenum concentrate is relatively high, sodium thioglycolate is added to inhibit copper. The copper content of molybdenum concentrate can be reduced from 0.5% to below 0.17%.
8. Oxalic Acid
The molecular formula of oxalic acid is (COOH)2 and its structural formula
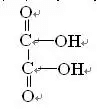
It is a saturated dibasic acid called oxalic acid, which is an organic inhibitor. Oxalic acid is an effective inhibitor of barite and garnet. In fluorite flotation, oxalic acid is often used to inhibit gangue minerals to ensure that the grade of CaF2 is 95% fluorite concentrate.
9. Carboxymethyl Cellulose
Carboxymethyl cellulose, an industrial product, is a light yellow flocculent substance with relatively stable chemical properties and is soluble in water.
Carboxymethyl cellulose is an effective inhibitor of magnesium-containing silicate minerals such as pyroxene, serpentine, hornblende, high amphitrite, and chlorite. It also has an inhibitory effect on quartz, carbonaceous gangue, and argillaceous gangue (weathering product of silicate minerals). The use of carboxymethyl cellulose to inhibit pyroxene and hornblende for nickel ore floatation is better than water glass.
pH regulators, inhibitors, activators, flocculants, and dispersants are generally called flotation regulators. Generally speaking, flotation agents can be divided into three categories: collectors, foaming agents, and regulators.
LATEST PRODUCTS
-
Cyclosizer
【Feeding Capacity】< 100g/time 【Feeding …
-
JKZ/2JKZ Mine Shaft Sinking Hoist
【Power Source】Electric Motor Drive 【Motor Spee…
-
JK(B)/2JK(B) Mine Hoisting Winch
【Capacity】 6-17 Tons 【Drum Number】1-2 【…









