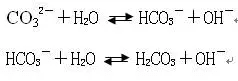In the process of beneficiation, taking advantage of the different natural hydrophobicity of minerals, the enrichment process of flotation of minerals from the grinding and classification overflow slurry is called flotation. In the flotation operation, in order to effectively separate the various minerals of the ground ore, it must be treated with chemicals, and the slurry should be stirred and aerated. The minerals that are easy to adhere to the bubbles float with the bubbles and do not adhere to the bubbles. The minerals remain in the pulp to achieve the purpose of mineral enrichment. The various agents used in the flotation process are collectively referred to as flotation agents. In the flotation agents, except for collectors and foaming agents, they are all called regulators. The role of the regulator is to adjust the role of the collector and the mineral, promote or inhibit the float-ability of the mineral; adjust the pH of the pulp and the composition of ions. According to the effect of regulators, it can be roughly divided into pH regulators, activators, inhibitors, dispersants, flocculants, etc. In this article, we will discuss pH regulators, activators, dispersants, and flocculants with you.

PH Regulator
PH regulators include lime, sodium carbonate, sulfuric acid, sulfur dioxide, caustic soda, etc.
1. Lime
Limestone (CaCO3) is calcined at 1200°C and decomposed into quicklime (CaO) and carbon dioxide. Quicklime is called lime for short. Quicklime is easy to absorb water to become hydrated lime (Ca(OH)2). Hydrated lime is a white powdery substance and is not easy to dissolve in water. It is usually added directly to the ball mill or the stirring tank before flotation during the flotation operation. It can also be adjusted into lime milk with water during stirring, and then added to the flotation machine. Calcium oxide is a strong base, and calcium hydroxide dissolved in water is completely ionized, making the solution strongly alkaline.
Lime is the cheapest slurry pH regulator. In polymetallic sulfide deposits, when priority flotation is used, lime is often used to increase the pH value of the slurry and inhibit pyrite. Lime is a typical inhibitor of pyrite. Generally speaking, some pyrite can be floated in weakly acidic pulp, and some can also be floated in the neutral or alkaline pulp. After the pyrite surface is oxidized, it will not float well when the pH is greater than 7. The addition of lime pyrite is suppressed.
The reason why lime inhibits pyrite is the formation of a hydrophilic film of Fe(OH)2 and Fe(OH)3 on the surface of the mineral.
For pyrite inhibited by lime, sodium carbonate and copper sulfate can be used, or sulfuric acid can be added to adjust the pH of the pulp to 6-7, and the pyrite can be floated again.
2. Sodium Carbonate
The scientific name of soda is sodium carbonate, and it is called soda ash in industry. It is a kind of weak acid and strong alkali salt, a colorless solid and easily soluble in water. Electrolysis in an aqueous solution is sodium ions and carbonate.
Na2CO3=2Na++CO32-
The carbonate is hydrolyzed again to make the solution alkaline
The sodium carbonate aqueous solution is weakly alkaline, and the pH value is between 8-10. In the selection of lead-zinc sulfide ore, priority flotation is used, galena is selected to suppress sphalerite and pyrite, and the pH of the slurry is adjusted to between 8-10. Using sodium carbonate instead of lime not only maintains the pH stability of the pulp but also avoids the inhibitory effect of Ca2+ on lead minerals.
3. Sulfuric Acid
The sulfuric acid used in industry is brown, the common concentrated sulfuric acid contains H2SO496%~98%, and the dilute sulfuric acid 63%~65%.
Sulfuric acid is the most commonly used acid regulator in flotation operations and concentrated sulfuric acid can be arbitrarily miscible with water. Sulfuric acid is a strong acid that ionizes large amounts of hydrogen ions in aqueous solutions.
H2SO4=2H++SO42-
When diluting and using, pay special attention to slowly injecting sulfuric acid into the water to avoid a large amount of heat from boiling the added water, causing sulfuric acid to splash and cause burns.
Activator
The effect of increasing the float-ability of minerals in the flotation process is called activation, and the agents used to change the composition of the mineral surface and promote the interaction of the collector with the mineral surface are called activators.
Activation can be roughly divided into 1. Spontaneous activation; 2. Pre-activation; 3. Resurrection; 4. Vulcanization.
1. Spontaneous Activation
When processing non-ferrous polymetallic ores, the surface of minerals and some soluble salt ions spontaneously during the grinding process, such as the symbiosis of sphalerite and copper sulfide minerals, the oxidation after the ore is mined There is always a small number of copper sulfide minerals that are oxidized to copper sulfate. Cu2+ ions in the slurry react with the surface of the sphalerite to activate it, which causes difficulties in the separation of copper and zinc. It is necessary to add lime or sodium carbonate and other adjusting agents to precipitate some may cause Activated “inevitable ion”.
2. Pre-activation
It means adding an activator to activate it in order to select a certain mineral. When the pyrite is heavily oxidized, add sulfuric acid to dissolve the oxide film on the surface of the pyrite before the pyrite is selected to expose a fresh surface to facilitate flotation.
3. Resurrection
The resurrection effect refers to a certain mineral that has been inhibited, such as sphalerite inhibited with cyanide, which can be resurrected by adding copper sulfate.
4. Sulfidation
Sulfidation means that the metal oxide ore is first treated with sodium sulfide to form a thin film of metal sulfide mineral on the surface of the oxide ore, and then flotation is carried out with xanthate.
The beneficiation reagents used as activators are Sulfuric acid, sulfurous acid, sodium sulfide, copper sulfate, oxalic acid, lime, sulfur dioxide, lead nitrate, sodium carbonate, sodium hydroxide, lead salt, barium salt, etc.
Flocculant & Dispersant
1. Flocculant
Flocculants are widely used in the flotation process. For example, in order to prevent the loss of concentrate and accelerate the sedimentation of fine concentrate particles in the concentrate thickener, flocculants should be added when necessary to reduce the loss of concentrate in the overflow of the thickener. Some concentrate slurry is difficult to filter. Adding flocculants can increase the filtration rate and reduce the moisture in the filter cake. It is more importantly used in the purification of tailings water to prevent the pollution of the tailings water to the surrounding environment, and at the same time create conditions for the recycling of tailings water.
Flocculants have been put into use: polyacrylamide, polyoxyethylene, and so on. The most widely used is polyacrylamide called No. 3 coagulant.
Flocculant No. 3 is a synthetic polymer compound. The production operation steps are divided into hydrolysis, neutralization, filtration, ion exchange, polymerization, and other processes. The shaping process for preparing polyacrylamide is sulfuric acid hydration, monomer aqueous solution polymerization, and the content of the finished polyacrylamide is 7% to 8%.
The dense filtration of scheelite using polyacrylamide agents can reduce the loss of concentrate in overflow. The raw ore contains 0.3%~0.6% WO3, the solid content in the overflow is 50~60g/L, and the content of WO3 0.6%~0.8%.
Polyacrylamide medicaments can also greatly accelerate the sedimentation rate of iron hydroxide, gypsum, scheelite, and barite.
In short, polyacrylamides have a wide range of uses and good effects. They can be used as gangue inhibitors, clay selective collectors, and good flocculants.
2. Dispersant
In flotation operations, the most commonly used dispersant is water glass. Water glass has a dispersing effect on the sludge and has a significant effect on improving the concentrate grade of valuable metals. The same agent often has different effects under different flotation conditions. Sodium silicate is a dispersant for dispersing sludge, an inhibitor for quartz, and it can also adjust the pH of the ore slurry and is a regulator. Therefore, the classification of medicaments has its flexibility. As far as water glass is concerned, it can be used as both a dispersant and an inhibitor.
LATEST PRODUCTS
-
Cyclosizer
【Feeding Capacity】< 100g/time 【Feeding …
-
JKZ/2JKZ Mine Shaft Sinking Hoist
【Power Source】Electric Motor Drive 【Motor Spee…
-
JK(B)/2JK(B) Mine Hoisting Winch
【Capacity】 6-17 Tons 【Drum Number】1-2 【…